 Notice how Na after in the second I.E, Mg in the third I.E., Al in the fourth I.E. Determine the number of protons, neutrons, and electrons in an atom. These are the ionization energies for the period three elements. 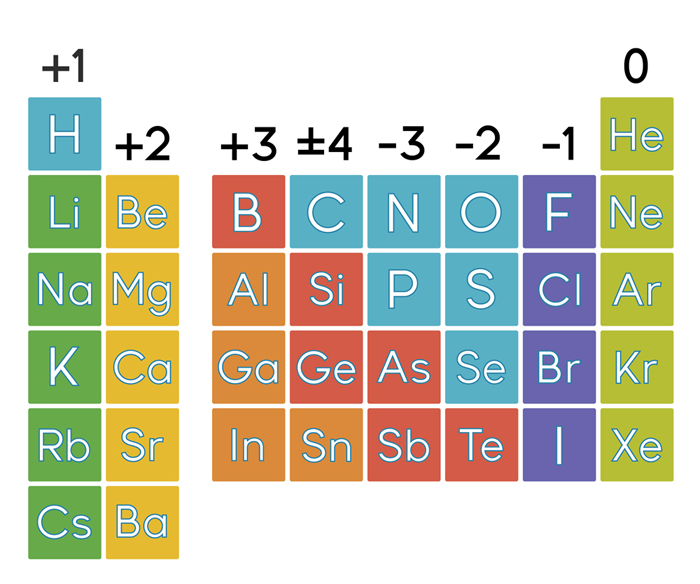
Large quantities of zinc are used to produce die-castings, which are important in the automobile, electrical and hardware industries. Galvanised steel is used for car bodies, street lamp posts, safety barriers and suspension bridges. These were some important difference between molecular mass and molar mass. Most zinc is used to galvanise other metals, such as iron, to prevent rusting. 
Example: Molecular mass of Ca (OH)2 74 atomic mass units. Therefore, it requires less energy to remove one of their valence electrons Table 1: Ionization Energies of certain elements (1st IE, 2nd IE, etc) Element Example: Mass of 1 mole of oxygen is 15.9994 grams. Additionally, elements in the left corner have a low ionization energy because losing an electron allows them to have the noble gas configuration. The highest amount of energy required occurs with the elements in the upper right hand corner. Note that some masses are in parentheses, usually towards the bottom of the table of elements, The parentheses basically mean that scientists don’t. Generally, this number is the one with the decimal point. If the mass of a substance is known, the number of moles in the substance can be calculated. The table has mass, or molar mass, shown as the number in the bottom of each square. The molar mass of an element is found on the periodic table, and it is the elements atomic weight in grams/mole (g/mol). The highest ionization energies are the noble gases because they all have high effective charge due to their octet formation and require a high amount of energy to destroy that stable configuration. Periodic Table with Mass Numbers and Atomic Symbols. 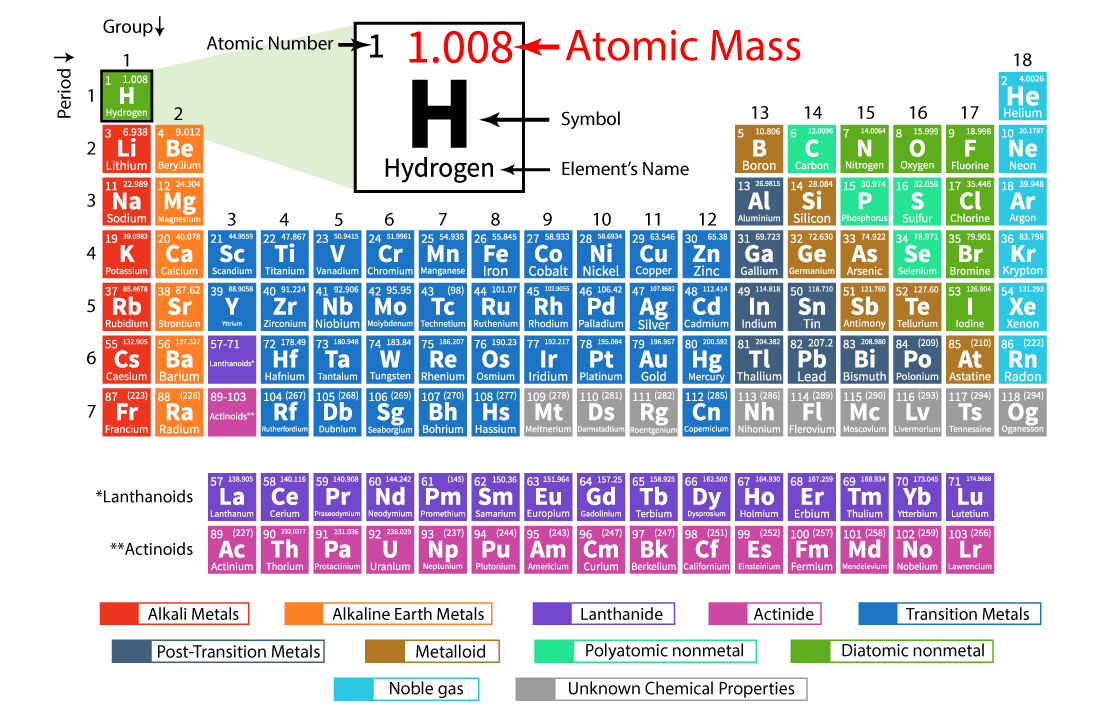
Ionization energies increase relative to high effective charge. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
